The non-ideal gas had a greater pressure by 1.602 atm. The pressure for the ideal gas is 30.55 atm and the pressure for van der Waals equation of the non-ideal gas was 32.152 atm. Introduzca el valor y haga clic en calcular para ver un paso a paso Van der Waals ley solución. Una forma de introducir todas las conocidas propiedades del gas y unidades serán presentados. 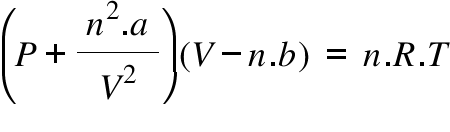
Part 3 - Find the difference between ideal and non-ideal conditions Seleccione una cantidad para resolver para y uno de los Van der Waals ley ecuaciones a utilizar. To make the math easier to follow, the equation will be broken into two parts where 
Van der Waals equation is expressed by the formulaĪ = attraction between individual gas particlesī = average volume of individual gas particles Use the Result Plot option to plot compressibility factor versus pressure and temperature, compressibility factor versus pressure and equation of state type, or compressibility factor versus temperature and equation of state type.The ideal gas law is expressed by the formula: Thus, intermolecular attraction effect reduces the pressure of the real gases. Hence, P (nRT/V - an2/V2)Pi Since, Pi nRT/V only. The compressibility factor calculation is valid for gas phase only. We have Van der Waals Equation, (P+ an2/V2) (V - nb) nRT but b0 (no size). The compressibility factor can be calculated from either the Peng Robinson, Soave, Redlich Kwong, or van der Waals equation of state (EOS). (i) Using Van der Waals equation, calculate the temperature of 20.0 mole of helium in a 10.0 litre cylinder at 120 atmosphere pressure. Help Using The Pipeng Toolbox (opens in the popup workbook) Links : ±ĬALCULATOR : Gas Compressibility Factor And Density From Cubic Equation Of State ±Ĭalculate gas molar volume, compressibility factor Z and density from the fluid pressure, temperature and critical point constants for selected gases. Try plus mode using the Plus Mode Demo tools with no login. Buy a Subscription to use the tools in plus mode (with plots, tables, goal seek etc). tools are free in basic CHECK mode with Login or Open a free account (CHECK values no plots, tables, goal seek etc). Login or Open a free account to use the tools in plus mode (with plots, tables, goal seek etc). tools are free in basic mode with no login (no plots, tables, goal seek etc). More Fluid Compressibility Z Factor Modules.You know its pressure P r, volume V r and temperature T and you wish to find the compressibility factor of this gas. Fluid Mixture From Kay's Rule Calculation Module The van der Waals equation is (2) ( P r + a n 2 ( V r) 2) ( V r n b) n R T Now, consider you have a container containing a 1 m o l real gas.Fluid Critical Point And Molar Mass Data Module.Fluid Compressibility Factor Data Module A Van der Waals Calculator can be used to calculate the strength of these forces and the resulting physical properties.Gas Compressibility Factor From The Cubic Equation Calculation Module.Gas Compressibility Factor Calculation Module.Reference : Kaye And Laby : Tables Of Physical And Chemical Constants Change Module : The gas mole volume is calculated by solving the quadratic equation, and the compressibility factor is calculated from the mole volume. The compressibility factor is calculated using the second order virial equation Gas Compressibility Factor From The Virial EquationĬalculate gas compressibility factor or Z factor from the virial equation. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
